Abstract
Non-Hodgkin lymphoma (NHL) is among the top 10 malignant tumors in incidence and mortality. Accumulating evidence indicates that obesity may influence the prognosis of several types of lymphoma, but the underlying mechanism is largely unknown for contributing to the poor prognosis or the aggressiveness of NHL. Obesity can result in abnormal lipid metabolism, especially abnormal sphingolipid metabolism. Existing data indicate the carcinogenetic role of Sphingosine-1-phosphate (S1P)/ sphingosine kinases (SPHK) signaling, we hypothesized that SIP/SPHK signaling played an important role in lymphomagenesis.
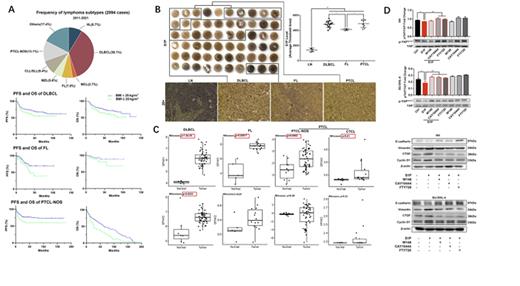
To test the hypothesis that obesity deteriorate the prognosis of NHL, we retrospectively investigated a cohort of 2094 patients who were diagnosed with lymphoma in the First Hospital of Jilin University from Jun 1st, 2011 to Jun 1st 2021. These patients were categorized into high Body Mass Index (BMI, defined as BMI≥25) group and low BMI (defined as BMI<25) group. The demographic information and imaging evidence of disease progression were extracted and analyzed. Survival data were follow-up till Jun 1st 2021. Progression-free survival (PFS) and overall survival (OS) of three main subtypes of lymphomas (namely Diffuse large B-cell lymphoma [DLBCL], follicular lymphoma [FL], peripheral T-cell lymphoma-not otherwise specified [PTCL-NOS]) in this cohort were compared with the time-to-event analysis. High BMI group had a negative impact on PFS and OS in all three groups (DLBCL [P < 0.05], FL [P < 0.05], and PTCL-NOS [P < 0.05]).(A) To investigate S1P signaling, we performed an IHC study to determine the levels of S1P in the tissue array-samples from various human lymphomas. By computer-imaging analysis, significantly increased S1P levels were detected in the tissues from DLBCL, follicular lymphoma (FL), and peripheral T-cell lymphoma (PTCL). Using a web-based database, Gene Expression Omnibus (GEO), the high throughput gene expression was profiled for SPHK1 and SPHK2 in the lymphomas (DLBCL, FL, and PTCL) and the respective normal controls. Significantly up-regulated expression of SPHK1 was found in DLBCL (n = 55) versus centroblast B-cells (n = 7), in FL (n = 14) versus normal B-cells (n = 5), and in PTCL (n = 80) versus normal T cells (n =10). Significantly up-regulated expression of SPHK2 were also detected in DLBCL but not in FL and PTCL in comparison with their respective normal controls.(B-C) To further investigate the potential mechanism of S1P-S1P receptors mediated lymphogenisis, we employed two human lymphoma cell lines (HH for T cell lymphoma and SU-DHL-4 for B cell lymphoma) for verification. The S1P mediated down-regulation of YAP phosphorylation was found in both HH cells and SU-DHL-4 cells. WB showed up-regulated S1PR1 and S1PR3, and down-regulated S1PR2, consistent with the qPCR results. Three S1PR inhibitors (W146 for S1PR1, CAY10444 for S1PR3, and FTY720 for S1PR1-5) were selected to confirm the hypothesis that S1P signaling could mediate the HIPPO pathway in lymphoma cells. XTT cell viability assay and a trans-well assay as well as WB were further performed to investigate cell proliferation, migration, and EMT. Consistently, blockage of S1PR1 and/or S1PR3 significantly inhibited the S1P induced cell proliferation and migration, down regulated the Cyclin D1 expression, and attenuated the alterations of E-cadherin/Vimentin in both HH cells and SU-DHL-4 cells. These results indicated that blockage of S1PR1 and/or S1PR3 significantly attenuated the S1P-induced decreases of YAP phosphorylation in both HH cells and SU-DHL-4 cells.(D) In conclusion, the NHL patients with obesity have a poor PFS and OS, which is associated with the elevated S1P-SPHK1 signaling. S1P-S1PR1/S1PR3-YAP signaling mediates lymphomagenesis contributing to tumor aggressive growth.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal